SMA: higher doses of nusinersen approved in Europe

The European Commission has just approved an extension for higher doses of nusinersen (Spinraza) in spinal muscular atrophy.
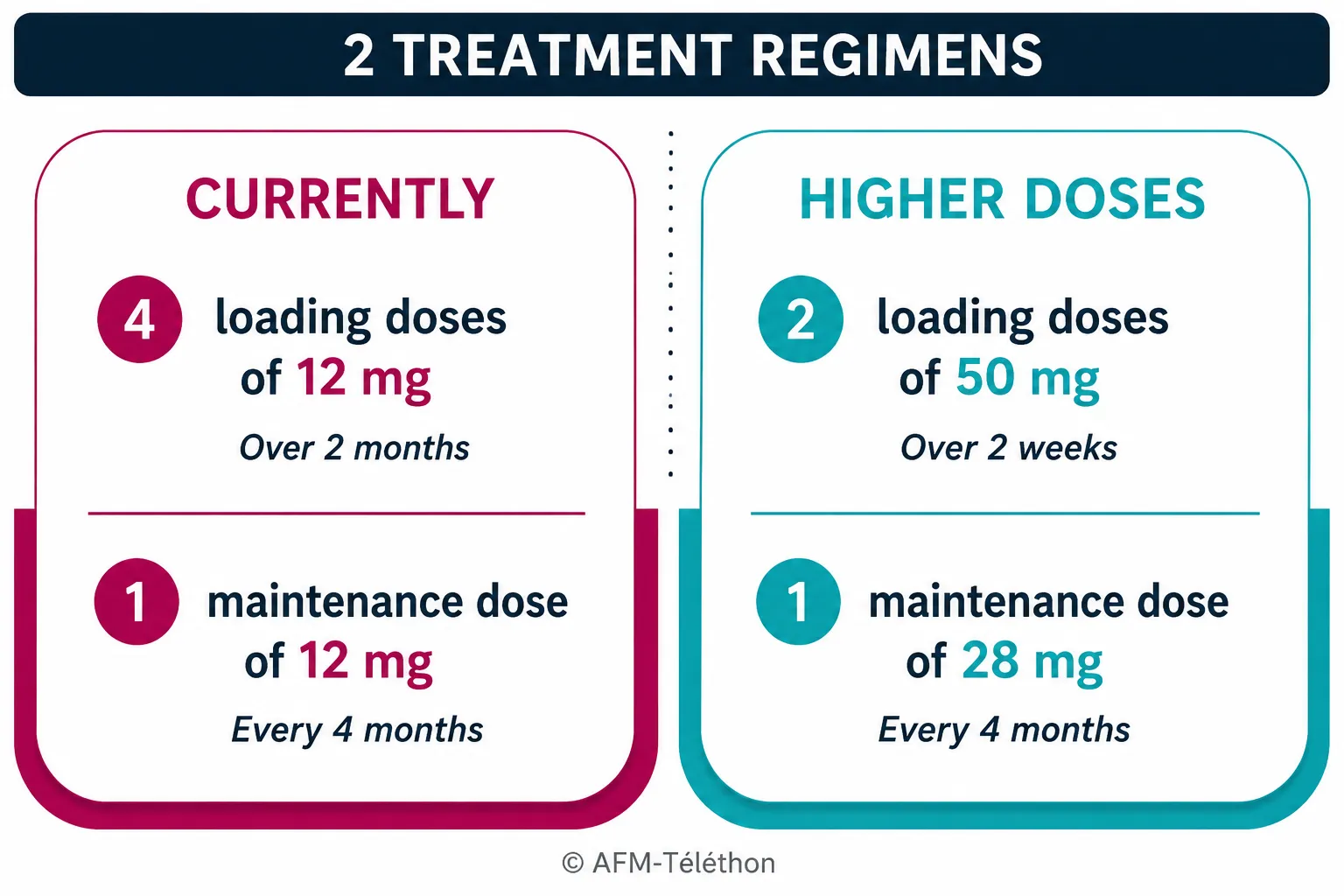
This European marketing authorisation is based on the results of the DEVOTE trial, which demonstrated beneficial effects of higher doses (50 mg and 28 mg) of intrathecal nusinersen in participants with spinal muscular atrophy (SMA), whether previously treated or not. These new doses were well tolerated and helped slow motor neuron degeneration and improve participants’ motor function. Because they are higher, they reduce by half the number of initial injections required (loading doses).
This new treatment regimen is added to the current protocol consisting of administering 12 mg doses intrathecally.
In France, health authorities must now validate these new dosing regimens before they become available. It will therefore take several months before patients can access them.
Source
Biogen Receives European Commission Approval for High Dose Regimen of SPINRAZA® (nusinersen) for Spinal Muscular Atrophy. Biogen. Press release, 12 January 2026.
